|
For example, fructofuranose fructose in a 5 membered ring. 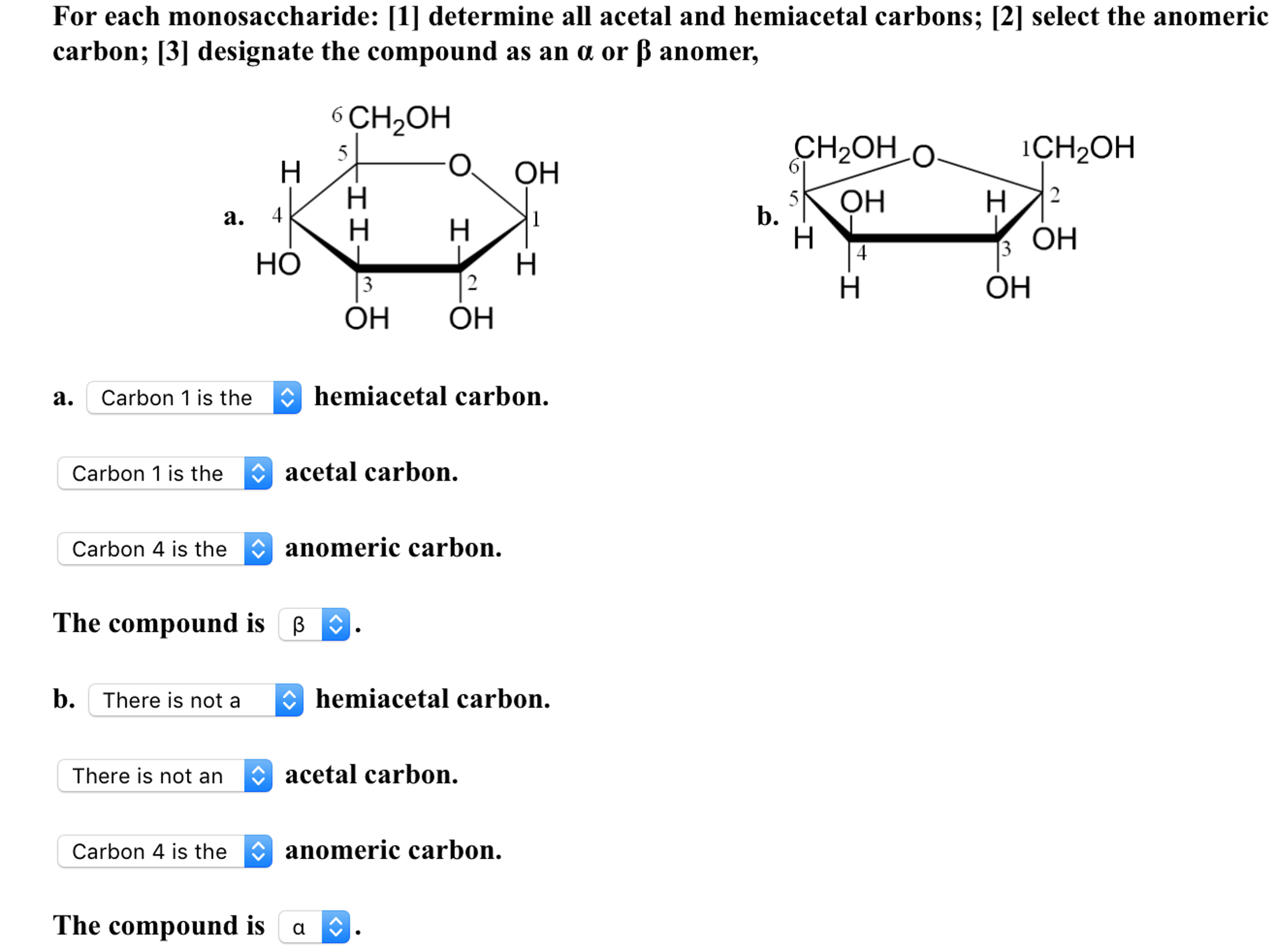
A sugar is in its -anomer form when the anomeric carbon’s substituent is on the opposite side of the plane as the highest numbered chiral center’s substituent. furanose sugars in a 5 membered ring structure pentagon shaped. Be aware that a cyclic sugar can exist in two possible anomers: an -anomer and a -anomer. Thus, all the molecules may eventually react, even though very little free aldehyde is present at a time. In a cyclic sugar, the anomeric carbon is the carbon that has two bonds to oxygen. how many chiral carbon atoms are present in fructose A carbon. As the small amount of free aldehyde is used up in a reaction, there is a shift in the equilibrium to yield more aldehyde. 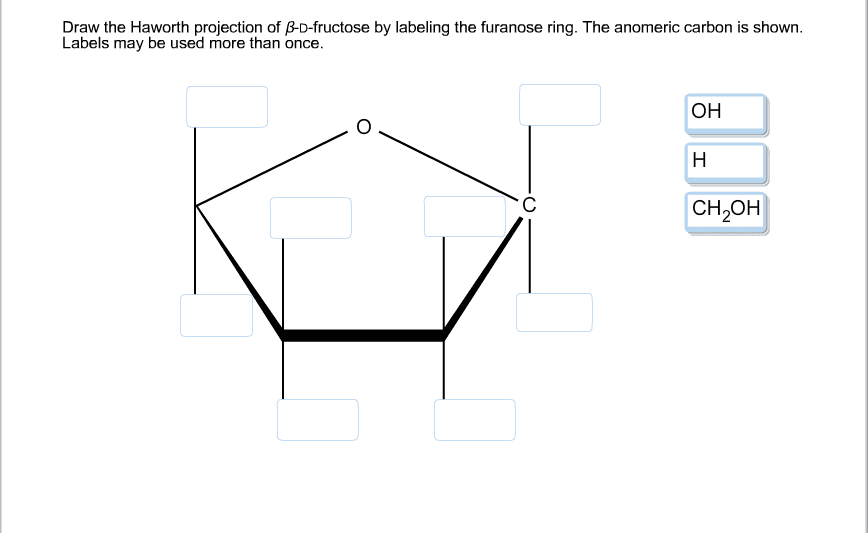
The interconversion between the forms is known as mutarotation, which is shown for D-glucose (a) and D-fructose (b).Įven though only a small percentage of the molecules are in the open-chain aldehyde form at any time, the solution will nevertheless exhibit the characteristic reactions of an aldehyde. Anomers are diastereoisomers of cyclic forms of sugars or similar molecules differing in the configuration at the anomeric carbon (C-1 atom of an aldose or the. In an aqueous solution, monosaccharides exist as an equilibrium mixture of three forms. The bond to the second monosaccharide (fructose) is tricky because the glycosidic bond is going to the anomeric carbon of fructose.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
